 
No patients died during the blinded phase of the RCTs. The most common AEs in the flumazenil group were agitation and gastrointestinal symptoms, whereas the most common SAEs were supraventricular arrhythmia and convulsions. SAEs were also significantly more common in the flumazenil group compared with the placebo group (12/498 versus 2/492 risk ratio: 3.81 95% CI: 1.28–11.39 p = 0.02). 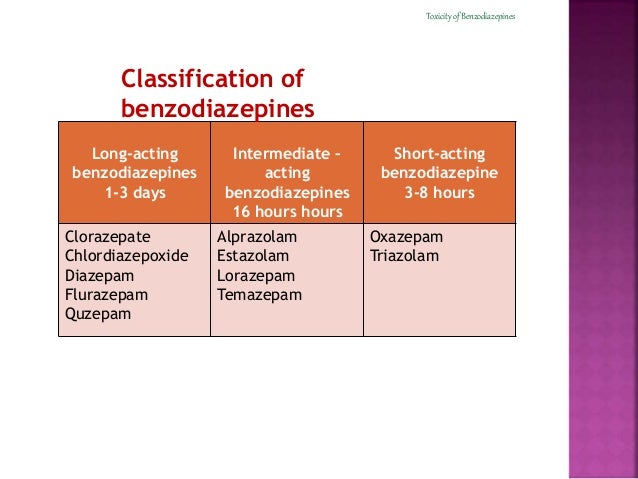 
AEs were significantly more common in the flumazenil group (138/498) compared with the placebo group (47/492) (risk ratio: 2.85 95% confidence interval: 2.11–3.84 p < 0.00001). Thirteen trials with a total of 994 randomised (990 evaluable) patients were included. Pre-defined outcome measures were AEs, SAEs and mortality. Randomised clinical trials (RCTs) in verified or suspected benzodiazepine overdose patients comparing treatment with flumazenil versus placebo were included. Studies included in the meta-analyses were identified by literature search in Medline, Cochrane Library and Embase using combinations of the words flumazenil, benzodiazepines, anti-anxiety agents, poisoning, toxicity and overdose. The objective was to assess the risk of (S)AEs associated with the use of flumazenil in patients with impaired consciousness due to known or suspected benzodiazepine overdose. Serious adverse events (SAEs) including seizures and cardiac arrhythmias have been reported in patients treated with flumazenil, and the clinical advantage of flumazenil treatment has been questioned. Flumazenil is used for the reversal of benzodiazepine overdose. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
